last modified: Sunday, 08-Nov-2009 04:44:32 CET
Document status: complete
Quartz is a compound of one part silicon and two parts of oxygen, silicon dioxide, SiO2. Its chemical composition (and the element silicon, Si) was discovered by the Swedish chemist Jöns Jakob Berzelius in 1823. Silicon dioxide is commonly called silica.
According to IUPAC, the following two names are correct: silicon dioxide, and dioxosilane[1].
|
10mm 1040x800 193kb - 2080x1600 684kb |
Reactions
At room temperature, SiO2 in all modifications is almost inert and does not react with most other substances. Even at moderately high temperatures silica is chemically very stable. For that reason fused quartz (silica glass) is widely used for chemical apparatus, especially when catalytic reactions of the metal cations in ordinary glass need to be avoided. The reason for the low reactivity of silica is the very strong Si-O bond, but also its macromolecular structure.Being the anhydrite of an acid itself (orthosilicic acid, H4SiO4), quartz will in general not be attacked by acids. The prominent exception is hydrofluoric acid[2], HF, which will decompose quartz to form first silicon fluoride SiF4, then hydrofluorosilicic acid:
SiO2 is also attacked by alkaline substances (like potassium hydroxide, KOH). The speed of the reaction depends on the modification and crystal size: Crystalline quartz will dissolve only very slowly in hot watery alkaline solutions, while amorphous SiO2, will be readily dissolved at room temperatures:
The reaction is of relevance for the collector, as alkaline solutions and detergents are sometimes used to remove moss and lichens from mineral specimen. Quartz crystals are usually not a problem, but I would be very careful when cleaning cryptocrystalline varieties.
A similar reaction leads to the formation of silica gels in concrete, when the alkaline compounds of the concrete, typically Ca(OH)2, react with amorphous silica and cryptocrystalline quartz (that is, opal and chalcedony). The silica gel attracts water, swells and cracks the concrete within a few decades.
All forms of silica dissolve in molten natron (Na2CO3) or potash (K2CO3) to form silicates:
At the high temperatures of many geological environments quartz acts as an acid and reacts with many alkaline minerals. A well known example is the formation of the mineral wollastonite Ca3Si3O9 from quartz and calcite in contact metamorphic processes at temperatures from about 600°C upwards:
The opposite reaction takes place at the weathering of silicate rocks, here the carbonic acid H2CO3 present, for example, in meteoric waters[3] releases silicic acid and forms carbonates[4].
Quartz is the "ore" of silicon. Silicon is retrieved at a temperature of about 2000°C in the following endothermic (energy-consuming) reaction:
The Si-O bond is much stronger than the C-O bond, and the reaction only works because carbon monoxide CO escapes from the system as a gas, so the system's equilibrium is pushed to the right side. The formation of carbon monoxide instead of carbon dioxide CO2 is typical for reductive reactions with carbon at high temperatures. The silicon formed in this process is not pure enough for chip production and needs to be purified in a rather complex procedure. So despite the abundance of its ore, pure silicon is relatively expensive.
Solubility in Water
The solubility of silicon dioxide in water is dependent on the temperature, the pressure, its surface structure, and on its structural modification[5]. The following table gives an overview of the solubility at room temperature and normal pressure.
| Substance | Solubility in Water at 25°C |
|---|---|
| Macrocrystalline Quartz | 2.9 mg/l [1] / 6-11 mg/l [2] |
| Chalcedony | 22-34mg/l [2] |
| Cristobalite | 6 mg/l [1] |
| Tridymite | 4.5 mg/l [1] |
| Stishovite | 11 mg/l [1] |
| Quartz Glass (amorphous) | 39 mg/l [1] / 120 mg/l [2] |
Data from:
[1] - ➛Hollemann & Wiberg, 1985
[2] - ➛Rykart, 1995
At room temperatures quartz is practically insoluble in water. Tap water is usually almost saturated with dissolved silica (with respect to quartz), and the dissolution process is very slow, so there is no need to worry about quartz crystals being damaged by repeated cleaning.
Nevertheless the dissolution of silica in bedrock and soils plays an important role in hot humid climates. The formation of laterite soils is linked to silica being gradually washed out of the surface layers and an enrichment of compounds that - at least under those climate conditions - are less soluble in water, like iron and aluminum oxides.
At temperatures above 100°C and high pressures the solubility of quartz increases quickly. At 300°C it is between 700 and 1200 mg/l, depending on the pressure.
SiO2 dissolves in water by forming orthosilicic acid, H4SiO4:
Orthosilicic acid is a very weak acid, weaker than carbonic acid, for example. It dissociates with a pK1 of 9.51 according to
There are no SiO2 molecules or aggregates of SiO4 tetrahedra present in a watery solution. The reversed reaction is the way quartz crystals grow, but a H4SiO4 molecule cannot simply dispose of 2 molecules of water, it needs another H4SiO4 molecule or an already present silica molecule with a hydroxyl (-OH) ending:
H4SiO4 + H4SiO4 → H2O + H6Si2O7 [9]
[SinOm]-OH + H4SiO4 → [Sin+1Om+2]-OH + 2 H2O [10]
Orthosilicic acid is only stable in a diluted watery solution, one cannot extract the pure acid. The most interesting property of orthosilicic acid is its tendency to polymerize in watery solutions to form first a sol and later a gel of polysilicic acids: orthosilicic acid molecules condensate to large molecules according to reactions [9] and [10]. As soon as you start to concentrate a H4SiO4 solution, the molecules will polymerize. At room temperature under laboratory conditions, the endpoint of this process is the formation of amorphous silica. Gels of polymerized silicic acid are probably also the predecessors of opal and cryptocrystalline quartz varieties (chalcedony in a wider sense) in nature.
Basic Structure of Silica
SiO2 occurs in no less than 13 different structural modifications. Their crystal structures (if present) might differ, but they all share some basic properties. The following applies to all silica modification except where noted. The molecular structure specific to quartz is covered in the chapter Structure.The chemical bonds in silica are covalent: they are based on molecular orbitals in which 2 electrons are shared between the atoms (as opposed to ionic bonds that are found in salts as fluorite or table salt; here electrons are transferred from one element to another to form ions of opposite electric charge, and in a crystal these ions are held together by electrostatic attraction).
|
.
|O|
- | -
.O-Si-O.
- | -
|O|
.
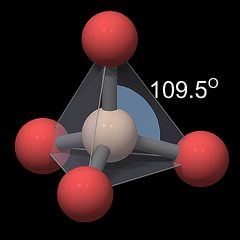
The geometry of this unit is shown in a ball-and-stick rendering in Fig.1, with silicon symbolized by an ivory colored, oxygen by a red sphere, and chemical bonds by gray pipes between them. The sizes of the atoms are not to scale. The oxygen atoms occupy positions around the silicon atom that keep them at the greatest possible distance from each other, so they form the four corners of a tetrahedron, symbolized by the translucent gray pyramid. Note that the oxygen atoms are not connected to each other, the gray lines between them have only been drawn to visualize the tetrahedral geometry. The angle of the central O-Si-O bonds is very close to the value in an ideal tetrahedron (109.5°, gray-blue arc in Fig.1), with a silicon oxygen distance of approximately 0.161 nm for all oxygen atoms, so the individual tetrahedron has an almost cubic symmetry[6].
The Si-O bond is highly polar, with electrons being more attracted by the oxygen, leading to an uneven distribution of electrical charges in the tetrahedron: the corners are more negatively, the center is more positively charged.
Individual molecules built like O=Si=O (with a linear structure like that of carbon dioxide CO2) can only be found in a SiO2 gas at temperatures above 2500°C.
Many textbooks explain the geometry of the SiO4-tetrahedron and other mineral structures as determined by the relative sizes of the different ions, as shown in Fig.2: The greater the relative size of the central ion, the more ions of opposite charge are necessary to enclose it. Accordingly, the SiO4-tetrahedron is often drawn as a small Si4+ cation enclosed by four large O2- anions (Fig.3). One can argue that this is inaccurate: Silicon and oxygen are connected by polar covalent bonds of joined electron orbitals, so they form a unit, and are not in the same way separated and freely movable as the ions in a salt. Compounds with Si4+ ions do in fact not exist[7]. There is some variation in the interpretation of the experimental findings, and while some authors view the Si-O bond as primarily covalent, others stress the ionic characteristics of the bond. For a review, see Gibbs et al., 1994 and Cohen, 1994.,
| |
O O
| |
-O-Si-O-Si-O-
| | | |
O O O O
| | | |
-O-Si-O-Si-O-Si-O-Si-O-
| | | |
O O O O
| | | |
-O-Si-O-
|
O
|
|
|
| Modification | Si-O-Si Angle | Si-O Distance / nm |
|---|---|---|
| α-Quartz[8] | 144° [1] 143.6°[3] |
0.16101- 0.16145 [2] |
| β-Quartz[8] | 153° [1] | 0.162 [1] 0.1609 [2] |
| α-Cristobalite | 147° [1] | 0.160-0.161 [1] |
| β-Cristobalite | 151° [1] | 0.158-0.169 [1] |
| α-Tridymite | ca.140° [1] | 0.154-0.171 [1] |
| β-Tridymite | 180° [1] | 0.153-0.155 [1] |
Data from:
[1] ➛Hollemann & Wiberg, 1985
[2] ➛Rykart, 1995
[3] Measurement with JMol based on unit cell coordinates.
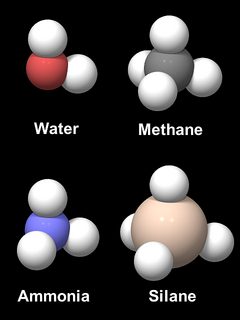
Oxygen has an inherent tendency to form an angular X-O-X bond, as do its neighbors in the periodic system of elements. Figure 6 shows models of four hydrogen compounds, water (H2O), methane (CH4), ammonia (NH3),and silane (SiH4). The inter-atomic distances and the relative sizes of C, N, O, and Si are to scale, but the hydrogen atoms are oversized for readability. The CH4 and SiH4 molecules are obviously tetrahedral. NH3 and H2O are almost shaped like a methane molecule with 1 or 2 hydrogen atoms removed, with an H-O-H angle of 104.5° and an H-N-H angle of 106.8°. The reason for this similarity is the geometry of the binding electron orbitals, the four so called sp3 hybrid orbitals, which form a tetrahedron. A tetrahedral arrangement of four sp3 hybrid orbitals is also found in silicon and is the main reason for the tetrahedral shape and the rigidity of the SiO4 unit in silica and silicates.
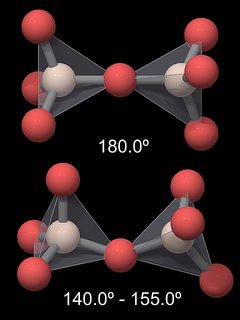
From this one would expect the Si-O-Si angle to be smaller, so why is it more open? For one, because the Si-O bond is highly polar and the oxygen atoms are negatively polarized, there are repulsive forces between the SiO4 groups, and a straight Si-O-Si would keep the oxygen atoms at greater distance. In addition, the length of the Si-O bond is shorter than calculated for a single bond: the value should be 0.181 nm but is in fact around 0.161 nm in quartz and has similar values in other silica modifications. This is an indication for a partial double binding character of this bond (Hollemann & Wiberg, 1985), which would also explain its great strength . This double bond is in accordance with the greater than expected Si-O-Si angle (144° instead of 109.5°). Note that the Si-O distance is shortest in β-tridymite, and this modification has a Si-O-Si angle of 180°. The theory behind the partial double binding character is fairly complex and I will not go further into it.
Silica and Silicates
Although chemically spoken silica is an oxide, in Angloamerican mineralogical literature quartz and all other silica modifications are usually classified as silicates (based on a classification scheme established by Dana). The reasoning behind calling quartz a silicate is that it is built up by a network of SiO4 tetrahedra. Silicates are classified according to the way SiO4 tetrahedra are integrated into the crystal structure and if and how the tetrahedra are linked to each other, and according to that quartz is called a tectosilicate ("network silicate" or "framework silicate").A silicate is a salt of silicic acids and one or several metals. Orthosilicic acid, H4SiO4, is just the simplest form, other silicic acids are made of several linked SiO4 units with attached hydrogen.
One could think that silicates form when a metal hydroxide and orthosilicic acid react, like
but this is not the way silicates usually form in nature. Silicates can be formed when a metal oxide or carbonate reacts with silica, like
Similar reactions do in fact occur in nature, the formation of wollastonite during metamorphic processes has already been mentioned (reaction [4]). But the normal way of silicate formation is that of a transformation: silicates react with each other to form new silicates. Once the SiO4 tetrahedron has formed, it is not easily broken apart, and can only be used in reactions to form new rings, chains, sheets, or networks of tetrahedra. Only very rarely and in very special environments can one find silicon in non-silicate minerals.
Purity of Quartz Crystals
Unlike most minerals, a clear quartz crystal is always chemically pure, with a SiO2-content approaching 99.5%, often more. There are basically two reasons for this.First, quartz has a macromolecular structure. It does not contain isolated ions of some element that could easily be replaced by another element. If, for example, a salt (a compound made of isolated ions held together by electrostatic forces) grows in a watery solution, its ions could be almost arbitrarily replaced as long as the size and charge of the replacing ions have an identical value. When quartz grows from a watery solution, there is literally nothing "attractive" on the growing surface to other ions in the solution. Only if, for example, an [AlO4]- group unit is built in instead of a SiO4 unit, the structure gets electrically imbalanced and allows an additional small monovalent cation (mostly H+, but also Na+ or Li+) to be included. Only a few elements fulfill the criteria to act as a replacement of silicon in the SiO4 unit, like aluminum in [AlO4]-, phosphorus in [PO4]+, or iron in [FeO4]- and trigger the inclusion of further ions. Interestingly, the mineral berlinite, AlPO4, which could be interpreted as the formula [Al,P]O2 is a structural isomorph of quartz, showing almost identical structural and symmetry properties, and very similar physical properties:
| Formula | Crystal System | Density | Hardness | Refractive Indices | |
|---|---|---|---|---|---|
| Berlinite | AlPO4 | trigonal | 2.64 g/cm3 | 7 | 1.524-1.530 |
| α-Quartz | SiO2 | trigonal | 2.65 g/cm3 | 7 | 1.544-1.553 |
Second, the three-dimensional network of SiO4 tetrahedra is quite rigid. Many minerals can tolerate relatively large amounts of certain other atoms and/or ions in their crystal lattice without showing great deviations in their physical properties[9]. The color might change completely, but the crystals appear clear and uniform, as the crystal lattice is somewhat flexible or spacious and maintains its original structure as a whole. The crystal lattice of quartz, however, is tight and very inflexible (making it hard but brittle), and only very small amounts of a number of other elements with small cations (usually Li, Na, H) can be built in during growth without distorting the structure of the lattice. And in fact the amount of monovalent cations found in quartz is roughly proportional to the amount of aluminum. Quartz that contains larger amounts of other substances is translucent to opaque, and often dull, because the crystal "grows around" the enclosed minerals, and light is scattered at the borders of the different materials.
This is not generally true for the other modifications of silica, and it seems that for some of them the presence of trace elements has in fact a stabilizing effect on their structure.
But while the overall composition of clear quartz crystals is remarkably pure (at least when compared to most other minerals), the impurities are typically not evenly distributed within the crystal. The concentrations of individual elements can vary by an order of magnitude within a few micrometers distance. For instance, the concentration of aluminum can vary between less than 5 ppma (parts per million atoms) to 10000 ppma within a single crystal (➛Perny et al. 1992). The pattern of the trace element distribution can be made visible with a technique called cathodoluminescence: high-energy electron beams ("cathode rays") induce short-lived luminescence effects that can be captured on ordinary film. The resulting photographs of thin slices of crystals show a fine laminar pattern. The layers all lie parallel to the crystal faces, similar to phantoms, so this pattern of trace element distribution is caused by rapid changes in the growth environment.
Chemically pure is still often not pure enough for technical purposes. A perfect, homogeneous crystal is as transparent as a vacuum, but trace elements cause distortions and defects in the crystal lattice, and this in turn effects the optical properties. Most of the elements built into quartz (H, Li, Al, Na, Ti, Fe) are ubiquitous and so further purification of silica is complicated and expensive. For that reason deposits of quartz with a low trace element content are still much sought after (see, for example, ➛Larsen et al. 2000).
Further Information, Literature, Links
A table of quartz properties can be found at mindat.org:http://www.mindat.org/min-3337.html
Another table at www.webmineral.com:
http://www.webmineral.com/data/Quartz.shtml
Footnotes
1 The IUPAC is an organization that aims at eliminating ambiguities and funny, but familiar names like Knallsäure (German for "banging acid", fulminic acid) from chemical nomenclature. Old names are still valid as long as they are unambiguous.
2
Some of you might find it tempting to do some experiments with hydrogen fluoride. Hydrogen fluoride is a very dangerous substance. Just look up a case in a good textbook on toxicology; after the lecture you will not want to touch it even with a pole. The watery solution is not a strong acid (pure HF is), and the undissociated small HF molecules easily penetrate the skin and bind calcium in the cells to form insoluble fluorite, CaF2. Calcium is essential for the live functions of all body cells. A small drop on a finger that goes unnoticed as it does not immediately hurt can - after some delay - cause substantial damage and great pain.
Let′s quickly do the math: 1 Mol HF = 20g. A drop (approximately 50 μl) of a 20% solution contains 0.00005 l x (200g/l) = 0.01 g. 10 mg, that doesn′t sound like much.
1 Mol Ca = 40g and 1 Mol F = 19g. 1 Ca will bind 2 F, thus 40g Ca will bind 38g F from 40g HF. 1 ml of you body tissue contains about 0.1mg Ca, and 0.1mg HF will let it precipitate almost completely. In theory, the 10mg HF in the small drop could precipitate most of the Ca in 100 ml tissue, killing all cells in it.
And no, I am not opposed to water fluoridation (no irony here).
3 The term "meteoric water" refers to its origin from the atmosphere, not from meteorites. The Greeks thought shooting stars came from the same sphere as the clouds: Stars and planets were considered eternal beings, something clouds and shooting stars are obviously not. Think of "meteorology".
4 That is why quartz in volcanic rocks is often accompanied by calcite.
5
This is somewhat counter-intuitive, but a saturated solution is in a thermodynamical equilibrium with the crystal. When a molecule is incorporated into a crystal structure, new bonds will be formed, and depending upon the geometry of the crystal structure, more or less energy is released or consumed. To dissolve a substance, energy is needed to break up the bonds, but energy is also released when water molecules orient themselves towards the dissolved molecule and form new electrostatic bonds (hydratisation energy). A more stable modification of a compound is generally less soluble, as more energy is required to break the bonds.
This has nothing to do with the speed of the reaction. Under certain conditions a more unstable modification will form first from a solution and only later it will be replaced by the stable modification: Cryptocrystalline quartz typically forms from a watery solution but with cristobalite and opal being intermediate steps, because the kinetics of the reactions are in favor of opal formation. And when you put a quartz crystal into 1 liter of pure water at room temperature, you can be pretty sure that when you take it out the next day it will not weigh 2.9 mg less. Probably it will be very hard to measure the difference. Maybe after a month, or after a year.
6 The tetrahedra in quartz are in fact not perfectly isometric like an ideal tetrahedron, but slightly distorted, because the distance to the central silicon atom is 0.16101 nm for 2 oxygen atoms, and 0.16145 nm for the 2 other oxygen atoms. This deviation from the ideal form is negligible for most practical purposes, however, as it is less than 0.3%.
7 The oxidation numbers of Si and O are indeed +4 and -2, respectively. The energy needed to actually remove 4 electrons from Si is too high to allow for a stable purely ionic bond. Even SiF4 is a compound with polar, but covalent bonds, despite the high electronegativity of fluorine. But Si4- anion compounds do exist (like Ca2Si).
8 Rykart (1995) gives the values 152.8° and 143.5° for α- and β-quartz, respectively. This seems to be a typographical error, the values should be switched.
9 But it′s not true that "anything goes". When a mineral forms, elements and ions are built into its crystal lattice when they meet certain criteria, like a specific charge, size, or electronegativity, and thus are similar to the ones that already form the lattice.
 Printer Friendly Version
Printer Friendly VersionCopyright © 2005-2013, A.C. A k h a v a n
Impressum - Source: http://www.quartzpage.de/gen_chem.html